Sapphire is chemically composed of aluminum oxide (Al₂O₃) and has a hexagonal crystal lattice structure. Sapphire crystals can be classified into two categories: natural sapphire and synthetic sapphire. In gemology and materials science, “sapphire” refers to all colored corundum crystals except red corundum. In other words, sapphire is not limited to blue aluminum oxide crystals alone.

In nature, sapphire crystals often contain trace impurity elements that give rise to different colors. When titanium ions (Ti³⁺) and iron ions (Fe³⁺) are present, the crystal appears blue and is referred to as blue sapphire. When chromium ions (Cr³⁺) are present, the crystal appears red and is known as ruby. When nickel ions (Ni³⁺) are present, the crystal may appear yellow and is referred to as yellow sapphire. Natural sapphire crystals are generally small in size and, after mining, are used primarily in jewelry and decorative applications.
Synthetic sapphire crystals, by contrast, are typically transparent and colorless because they do not contain chromophore ions. They are produced by placing amorphous high-purity alumina raw materials into a crucible and using a controlled crystal growth process. Under high temperature, the alumina is melted and then recrystallized, forming synthetic sapphire crystals with specific shapes and dimensions.
Synthetic sapphire exhibits excellent physical and chemical properties, which make it suitable for a wide range of applications.
Sapphire’s chemical stability is exceptionally high. It resists corrosion in almost all chemical environments.
Its mechanical properties are equally outstanding. Sapphire offers high hardness and high strength, maintaining excellent wear resistance, structural stability, and mechanical performance across environments ranging from cryogenic temperatures up to 1500°C. It is currently the hardest known oxide crystal material, with a Mohs hardness of 9, second only to diamond (Mohs 10).
Sapphire also possesses remarkable optical properties. The transmission wavelength range of sapphire single crystals extends from 0.19 μm to 5.5 μm. Double-side polished sapphire windows can achieve visible light transmittance above 86%, making sapphire widely applicable in the design and manufacture of optical window products.
In addition, sapphire’s dielectric constant is approximately 9.3 perpendicular to the C-axis and 11.5 parallel to the C-axis, roughly twice that of glass. Touch displays fabricated with sapphire substrates therefore offer very high sensitivity.
Sapphire windows made from synthetic sapphire can achieve a four-point bending strength of over 1000 MPa, more than 1.5 times that of conventional glass products. Moreover, because sapphire’s thermal conductivity is about twice that of glass, its heat dissipation rate is also approximately twice as high.
01
Growth Methods for Synthetic Sapphire Crystals
The main methods used for synthetic sapphire crystal growth include the flame fusion method, Czochralski pulling method, zone melting method, edge-defined film-fed growth (EFG) method, moving crucible method, heat exchange method, temperature gradient method, and Kyropoulos method, among others.
As sapphire crystal growth technology continues to advance, the methods that best meet market demand will be those characterized by a high degree of automation, minimal manual intervention, stable processes, consistent yield, reliable production capacity, and relatively low cost. These features define the future direction and mainstream trend of sapphire crystal growth technology.
Among the many growth methods available, only the Heat Exchange Method (HEM) and the Kyropoulos Method (KY) are capable of producing relatively large synthetic sapphire crystals.
Below is an introduction to these two internationally mainstream sapphire crystal growth methods.
1.1
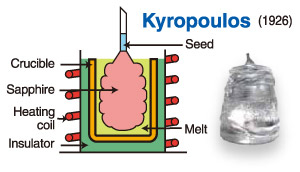
Kyropoulos Method
The Kyropoulos method, abbreviated as the KY method, was first proposed by Kyropoulos in 1926 for crystal growth. For a considerable period afterward, it was mainly used in the preparation and study of large halide crystals, hydroxides, carbonates, and similar materials.
Its principle is similar to that of the Czochralski method. First, the raw material is heated above its melting point to form a molten bath. A seed crystal is then brought into contact with the melt surface, initiating single-crystal growth with the same crystal structure as the seed at the solid-liquid interface. The seed is slowly pulled upward. After a certain pulling distance, a crystal neck is formed. Once the solidification rate at the seed-melt interface becomes stable, the pulling and rotation stop. The crystal is then grown mainly by controlling the cooling rate, allowing the single crystal to solidify gradually downward from the top until the entire ingot is formed.
In the 1960s and 1970s, the Kyropoulos method was improved and applied to the production of sapphire single crystals. Sapphire crystals grown by this method are typically pear-shaped. Their diameter can reach about 300 mm, their height can reach about 600 mm, and their weight is generally around 100 kg.
The main characteristics of sapphire growth by the Kyropoulos method are as follows:
- Manual seeding and greater operator dependence
The seeding process relies heavily on manual operation, requiring highly skilled technicians. The growth process also involves considerable operator judgment and intervention, resulting in a relatively low level of automation. Crystal yield may fluctuate significantly depending on personnel changes. - Fewer grain-boundary defects
During growth, the crystal does not contact the crucible wall, so grain-boundary defects are relatively limited. - Higher probability of microbubbles
Crystal growth occurs in a suspended state against gravity. As the crystal is rotated and pulled upward, microbubbles are more likely to form. As a result, crystals grown by this method generally have lower density than those produced by static sapphire growth methods, and the probability of bubble formation is relatively high. - Irregular crystal shape and lower material utilization
The grown crystals are non-cylindrical and irregular in shape, resulting in relatively low finished-product utilization. - Shorter service life of the thermal field system
The thermal field typically uses high-temperature-resistant metallic materials. Compared with inorganic materials such as graphite thermal fields, these components generally have shorter operating life and fewer reuse cycles.
1.2
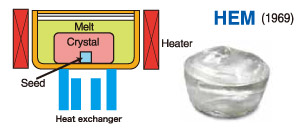
Heat Exchange Method
The Heat Exchange Method (HEM) is a sapphire crystal growth technology invented specifically for producing large sapphire crystals. It was jointly developed by Schmid and Viechnicki in the United States.
Its working principle is based on removing heat through a heat exchanger, thereby creating a vertical temperature gradient in the crystal growth zone, with lower temperature at the bottom and higher temperature at the top. By controlling the flow rate of helium gas in the heat exchanger—helium being a gas with high thermal conductivity that efficiently removes heat—and adjusting the heating power, this temperature gradient can be precisely regulated. As a result, the molten alumina inside the crucible solidifies upward from the seed crystal located at the bottom, forming a synthetic sapphire crystal.
This technology was later acquired by GT, a specialized U.S. crystal furnace manufacturer. Through systematic research and development, it became a patented technology used in GT’s automated sapphire crystal growth furnaces. After being applied and developed in the United States, the technology was introduced to China only in recent years, where it quickly gained recognition in the market and among industry peers.
Because the HEM process was specifically developed for the production of large sapphire crystals, it offers high automation, large production capacity, and stable yield, making it highly suitable for large-scale industrial manufacturing. It represents one of the most important directions and market demands in the current sapphire crystal materials industry. Crystals grown by this method can reach diameters of up to 600 mm, heights of up to 550 mm, and weights exceeding 300 kg.
The main characteristics of the Heat Exchange Method are as follows:
- Highly automated production process
The entire process—from raw material melting, seeding, and temperature gradient formation to crystal growth and annealing—is controlled by computer programs. No manual intervention is required, so production automation is high and yield does not fluctuate with staffing changes. - Contact with the crucible wall
During growth, the crystal remains in contact with the crucible wall, which makes grain-boundary defects more likely to occur along the crystal periphery. - Static bottom-up growth with high crystal quality
Crystal growth proceeds in a static, bottom-up manner. External disturbances during growth are relatively small, resulting in crystals with better density and hardness than those produced by non-static growth methods. Bubble density is also relatively low. - Customizable crystal dimensions
HEM allows sapphire crystals to be grown in customized diameters and heights. The crucible shape and size can be designed according to the final product dimensions, thereby maximizing material utilization. - High process complexity and cost
The method requires highly precise equipment and a complex process flow. Crystal growth cycles are long, helium is required as a coolant, and the crucible is single-use, resulting in relatively high production costs. - Regular crystal shape and suitability for mass production
The growth process produces crystals with uniform diameter, regular geometry, and relatively large size. Material utilization is high, yield is stable, and output is consistent, making the method especially suitable for large-scale manufacturing.
02
Conclusion
Overall, the development of high-quality, low-cost sapphire crystal growth technology remains a major focus and technical challenge worldwide. At the same time, large-size sapphire is clearly the mainstream direction for future crystal development. In this context, the Heat Exchange Method is particularly well suited to the needs of consumer electronics products such as smartphones.
2.1
Key Requirements of Consumer Electronics
Consumer electronics products such as smartphones have the following demand characteristics:
- Large overall demand, requiring suppliers to provide high production capacity and stable delivery
- Orders are often urgent, requiring the ability to deliver in a short time
- Strict requirements on microbubble density within sapphire crystal materials
- Crystals must have regular shape, complete geometry, and relatively large dimensions to support product design and manufacturing
2.2
Advantages of the Heat Exchange Method
Compared with other sapphire growth methods such as the Kyropoulos method and EFG-type molding methods, the Heat Exchange Method offers the following advantages:
- High degree of automation, low process fluctuation, stable yield, and reliable production capacity
- Large crystals with complete and regular geometry, suitable for a wide variety of product designs and manufacturing needs
- Static crystal growth process, resulting in higher crystal density, greater hardness and wear resistance, and lower microbubble density within the crystal
2.3
Why HEM Best Fits Consumer Electronics
Because of the above advantages, the Heat Exchange Method is the most suitable scalable crystal growth method for the consumer electronics industry, especially for products such as smartphones. Apple’s substantial investment in comparative research on various sapphire growth methods, and its eventual preference for the Heat Exchange Method, further supports this conclusion.
03
Closing Remarks
There are multiple methods for growing synthetic sapphire crystals. From the perspective of product application, sapphire ingots grown by the Kyropoulos method tend to have relatively irregular shapes and are therefore more suitable for machining into sapphire rods, making this method better suited for sapphire LED substrate applications.
By contrast, sapphire crystals grown by the Heat Exchange Method have regular shapes, larger diameters, and greater mass. In addition, the process features a high degree of automation, stable production capacity, and consistent yield. These characteristics enable the production of sapphire smartphone cover panels of 5 inches and above. For this reason, the Heat Exchange Method is more suitable for consumer electronics applications such as smartphones.
